-
WHAT IS NASA PHYSICS?
-
MODULES
-
Forces and Motion
-
Conservation of Momentum & Energy
-
Temperature and Heat
-
Fluids
-
Optics
-
Electromagnetic Spectrum
-
Modern Physics
-
Anticipation Guide 7
-
Intro to Modern Physics
-
Blackbody Radiation
-
The Ultraviolet Catastrophe
-
The Photoelectric Effect
-
Bohr's Atom
-
Spectra
-
Radioactive Decay
-
Special Relativity (SR)
-
Simultaneity
-
Distance and Time
-
General Relativity
-
May the Forces be with You
-
Modern Physics Notebook
-
Assessment Problems 7
-
-
Useful Things
-
-
SITE MAP
Temperature and Heat
-
Anticipation Guide
-
Thermal Energy
-
Measuring Temperature
-
Heat Expansion
-
Heat
-
Heat Capacity
-
Change of State
-
Transferring Heat
-
Greenhouse Effect
-
Notebook
-
Assessment Problems
The heat of fusion is the amount of heat needed to convert 1 g of a solid at its melting point into a liquid.
Equation
Q = mHf
3.9
Equation
Q = mHv
3.10
Change of State
 No, we don’t mean moving from New York to Texas. State and phase are words used interchangeably in physics to describe whether a material is a solid, liquid, gas or plasma – more on plasmas later. When heat is added to a material, at some point the material changes its state. Warming ice to 0°C causes it to change to water, a solid to liquid phase change. Continuing to add heat causes the water’s temperature to rise, ultimately reaching 100°C and the liquid begins to evaporate or become a gas.
No, we don’t mean moving from New York to Texas. State and phase are words used interchangeably in physics to describe whether a material is a solid, liquid, gas or plasma – more on plasmas later. When heat is added to a material, at some point the material changes its state. Warming ice to 0°C causes it to change to water, a solid to liquid phase change. Continuing to add heat causes the water’s temperature to rise, ultimately reaching 100°C and the liquid begins to evaporate or become a gas.
Left: Condensation; Image from Wikipedia
And just as a person might move back from Texas to New York, materials can change their states in the opposite direction when they cool. Water condenses from a gas to a liquid – you see that all the time when droplets of water form on a cold bottle of water just brought out of a refrigerator into the afternoon sunlight. To make water colder you can fill an ice cube tray with water, put it in the freezer, and the liquid will solidify as heat is removed. We are familiar with these state changes, and with physics you can calculate how much energy is required to change states. And you will also learn unexpected things like why boiling water can freeze almost instantly into ice.
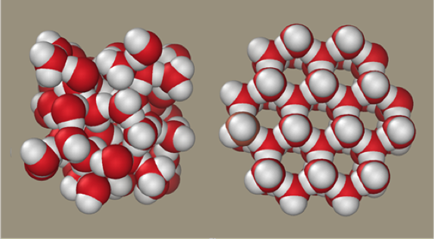 H2O; Image from WikipediaSubstances change state because of the way their atoms and molecules react to heat. Consider water, which is a simple molecule made of one atom of oxygen (red in diagram) and two atoms of hydrogen (white). Molecules in a liquid (left) are closely bonded, whereas molecules making up ice (right) are interconnected in a more open but rigid structure of six-sided ice crystals. Each hydrogen atom is connected to two oxygen atoms, and a strong regular structure results.
H2O; Image from WikipediaSubstances change state because of the way their atoms and molecules react to heat. Consider water, which is a simple molecule made of one atom of oxygen (red in diagram) and two atoms of hydrogen (white). Molecules in a liquid (left) are closely bonded, whereas molecules making up ice (right) are interconnected in a more open but rigid structure of six-sided ice crystals. Each hydrogen atom is connected to two oxygen atoms, and a strong regular structure results.
Heating ice causes its temperature to rise and its molecules to vibrate, in a sense pulling the electrons away from the oxygens. But the strength of the bonds is great so the molecule stays connected to other molecules. When the temperature reaches 0°C enough energy has been introduced into the ice to break the bonds connecting molecule to molecule, and the individual molecules become free to move. The solid ice begins to break apart, forming water. If energy continues to be added to the ice-water mixture the temperature does not change - its stays at 0°C - until all the ice is melted. All of the added energy goes to breaking the tight bonds of ice.
Additional heating of water causes its temperature to increase because the water molecules move more rapidly, until the temperature reaches 100°C. What happens then? At 100°C the water begins to boil, which means that the water molecules are breaking apart and steam (hot water vapor) is created. Ice, liquid water and steam all have the same chemical formula of H2O, but the H2O has very different properties in its different phases.
Energy to Change States
It takes exactly 80 calories of heat to turn 1 g of ice into 1 g of liquid water. This is called the heat of fusion of water.
A liquid releases the same amount of heat when it solidifies. Note that while a solid melts to become a liquid, or a liquid freezes to become solid, there is no increase in temperature because all of the energy goes into the phase change. Different materials require differing amounts of heat to melt.
If you want to melt 100 g of ice you would need 80 calories of heat for each gram or 8,000 calories for the entire amount of ice. Why is this type of information important? Well, if you were operating a factory and had to melt solid iron to pour into molds to make plows, for example, you would probably want to calculate how much energy was required so you could estimate the cost. Iron is easier to melt than ice, requiring only 65 calories for each gram. To melt 1,000 g of iron already at its melting temperature would require
65 calories/g x 1,000 g = 65,000 calories of energy.
Of course, first you would have to heat the iron to its melting point of 1,535°C before it would melt – and that would take additional energy!
Lets go back to water and trace its energy use as it changes from ice to liquid to steam. The animation summaries the critical temperatures and heat required for 1 gram throughout all the phases.
Here is a table that summarizes all this:
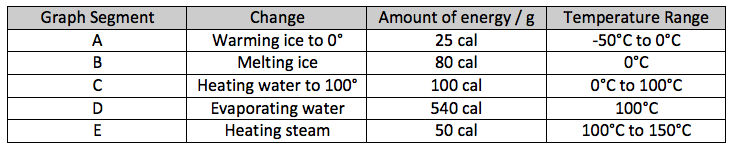
A: The specific heat of ice is 0.5 so to heat the ice from its starting temperature, say -50°C to its melting temperature of 0°C requires 0.5 cal/degree x 50 degrees = 25 calories per gram of ice.
B: As we saw earlier, to melt ice requires 80 calories/g, with the temperature of the water-ice mixtures staying constant at 0°C until all the ice has melted.
C: To heat each gram of water from 0°C to 100°C requires 1 calorie of energy for each degree of warming or a total of 100 calories/g for the 100°C increase.
D: Once the water reaches 100° the temperature remains constant as it starts to boil – or vaporize – and this requires 540 calories for each gram. This is much more than melting ice or heating water, because now the bonds between each water molecule are being broken, rather than just being stretched and reorganized.
E: After all the water has become a gas – steam – the addition of more heat will raise the temperature of the gas, that is, make the gas molecules move faster.
To cool a gas to liquid and then to an ice requires removing energy from the water for each step. For example, to cool water from 100°C to 0°C requires the water to lose 100 calories for each gram. The heat removed usually goes into the surrounding air. Another example: a wet T-shirt chills the wearer because 540 calories of body heat are removed to evaporate each gram of water from the shirt. On a hot day this is a cheap way to keep cool, but is generally not approved for classroom application.
To calculate the energy needed to melt a solid already at its melting temperature use this equation:
Heat needed = mass in grams x heat of fusion
Q = mHf
Where Hf is the heat of fusion of the solid.
To calculate the energy needed to vaporize a liquid already at its evaporation temperature use this equation:
Heat needed = mass in grams x heat of vaporization
Q = mHv
Where Hv is the heat of vaporization of the liquid.
Crystalline and Amorphous Solids
 Water ice is a crystalline solid. All of the molecules are linked together with the same type of bonds. Once the ice’s temperature reaches 0°C all of the bonds begin to break and melting occurs. Some other solids don’t have regular crystal structure and do have chemical bonds of different types, so that melting occurs over a range of temperatures as each type of bond breaks. Such substances are called amorphous solids. Two of the most familiar examples are butter and glass. We all know that butter gradually softens when warmed and as the heat increases it finally completely melts to a sticky liquid. Although we don’t often get to see glass melt it behaves in the same manner. In both cases the melting occurs over a range of temperatures rather than at one temperature as with a crystalline solid.
Water ice is a crystalline solid. All of the molecules are linked together with the same type of bonds. Once the ice’s temperature reaches 0°C all of the bonds begin to break and melting occurs. Some other solids don’t have regular crystal structure and do have chemical bonds of different types, so that melting occurs over a range of temperatures as each type of bond breaks. Such substances are called amorphous solids. Two of the most familiar examples are butter and glass. We all know that butter gradually softens when warmed and as the heat increases it finally completely melts to a sticky liquid. Although we don’t often get to see glass melt it behaves in the same manner. In both cases the melting occurs over a range of temperatures rather than at one temperature as with a crystalline solid.
Left: Butter sculpture of astronaut Neil Armstrong. Neil is displayed in a chilled space to avoid disastrous phase change. Image from Flickr.
© 2013 by Wheeling Jesuit University/Center for Educational Technologies®. 316 Washington Ave., Wheeling, WV 26003-6243. All rights reserved. Privacy Policy and Terms of Use.

