-
WHAT IS NASA PHYSICS?
-
MODULES
-
Forces and Motion
-
Conservation of Momentum & Energy
-
Temperature and Heat
-
Fluids
-
Optics
-
Electromagnetic Spectrum
-
Modern Physics
-
Anticipation Guide 7
-
Intro to Modern Physics
-
Blackbody Radiation
-
The Ultraviolet Catastrophe
-
The Photoelectric Effect
-
Bohr's Atom
-
Spectra
-
Radioactive Decay
-
Special Relativity (SR)
-
Simultaneity
-
Distance and Time
-
General Relativity
-
May the Forces be with You
-
Modern Physics Notebook
-
Assessment Problems 7
-
-
Useful Things
-
-
SITE MAP
Modern Physics
-
Anticipation Guide
-
Intro to Modern Physics
-
Blackbody Radiation
-
The Ultraviolet Catastrophe
-
The Photoelectric Effect
-
Bohr's Atom
-
Spectra
-
Radioactive Decay
-
Special Relativity (SR)
-
Simultaneity
-
Distance and Time
-
General Relativity
-
May the Forces be with You
-
Notebook
-
Assessment Problems
In modern physics the theories of general and special relativity describe the behavior of the large scale universe and very high velocities, and quantum physics describes the behavior of the small scale particle world.
Q: Why can't you trust an atom?
A: They make up everything
A neutron walked into a bar and asked, "How much for a drink?" The bartender replied, "For you, no charge."
Intro to Modern Physics

Above: Giants of modern physics and their mustaches in 1927; Einstein, front and center; Image from Wikipedia.
For almost 200 years, the field of physics relied on the seemingly irrefutable theories of force and motion put forth by Sir Isaac Newton in the 1600’s. Newtonian physics had been tested time and time again and accurately describes the observable universe. Orbital motions, the force of gravity, momentum, energy, all seemed perfectly determinant and quantifiable under these laws of what is today known as “classical physics”. But starting in the early 1900s, physicists discovered that classical physics fails to represent what happens at the extremes of size and speeds. For interactions at sub-atomic size or near the speed of light, Newtonian physics falls apart. Fortunately, Einstein and other brilliant scientists developed what is called “modern physics”, new, often non-intuitive ways to understand physics in the extremes. Modern physics – now more than 100 years old! – is not finished, and physicists are still trying to find a single theory that accurately describes everything at all sizes and velocities. This Theory of Everything does not yet exist – perhaps you will help create it.
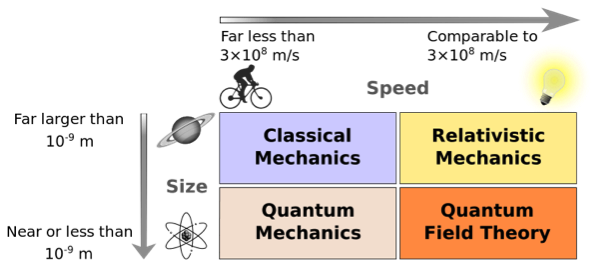 This graph shows how size and speed determine which types of physics hold. Classical (or Newtonian) mechanics describe the everyday world. Things that move near the speed of light (3 x 108 m/s) require relativistic mechanics developed by Einstein and others. Quantum mechanics is concerned with atomic size forces and energy, and quantum field theory is for things that are both very small and very fast.
This graph shows how size and speed determine which types of physics hold. Classical (or Newtonian) mechanics describe the everyday world. Things that move near the speed of light (3 x 108 m/s) require relativistic mechanics developed by Einstein and others. Quantum mechanics is concerned with atomic size forces and energy, and quantum field theory is for things that are both very small and very fast.
Above: Physics graph; Image from Wikipedia.
What Are Things Made Of?
 Democritus; Image from WikipediaModern physics is based on the atom, which had been speculated on for nearly 2500 years. In about 400 BC the Greek philosopher Democritus proposed that all matter is made of tiny, indivisible particles that he called atoms. He had no evidence that atoms existed, and no one else did for nearly 2000 years, but it seemed reasonable that big things were made of smaller things. In the late 1600s Isaac Newton had essentially the same concept of an atom as a tiny indestructible sphere. The kinetic theory of gases, which developed in the 1700s described atoms as small particles that moved around at various speeds, completely consistent with Democritus’ idea.
Democritus; Image from WikipediaModern physics is based on the atom, which had been speculated on for nearly 2500 years. In about 400 BC the Greek philosopher Democritus proposed that all matter is made of tiny, indivisible particles that he called atoms. He had no evidence that atoms existed, and no one else did for nearly 2000 years, but it seemed reasonable that big things were made of smaller things. In the late 1600s Isaac Newton had essentially the same concept of an atom as a tiny indestructible sphere. The kinetic theory of gases, which developed in the 1700s described atoms as small particles that moved around at various speeds, completely consistent with Democritus’ idea.
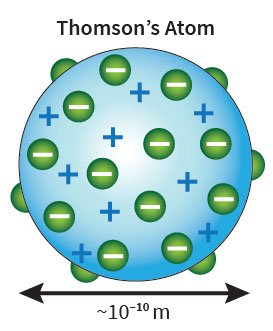 But the discovery of electrons in 1897, made it clear that atoms were more complicated, that they had parts and were divisible. Joseph John Thomson, a British physicist who discovered electrons, proposed that atoms were spheres of positive material with embedded pieces of negative charges, electrons, so that adding it all together atoms were neutral. Thomson’s model was similar to a spherical chocolate chip cookie, with the dough being positive and the chips being isolated bits of negative charge.
But the discovery of electrons in 1897, made it clear that atoms were more complicated, that they had parts and were divisible. Joseph John Thomson, a British physicist who discovered electrons, proposed that atoms were spheres of positive material with embedded pieces of negative charges, electrons, so that adding it all together atoms were neutral. Thomson’s model was similar to a spherical chocolate chip cookie, with the dough being positive and the chips being isolated bits of negative charge.
 That was a good model until 14 years later when Thompson’s student, Ernest Rutherford conducted an experiment that showed that all the positive charges were concentrated at the center of an atom, and not spread out as Thomson proposed. Rutherford called the concentrated positive charge the nucleus of an atom and compared the electrons that swirled around the nucleus to planets in orbit around the Sun. Both Thompson and Rutherford received Nobel prizes for their work, and Thompson was even knighted – Sir Joseph.
That was a good model until 14 years later when Thompson’s student, Ernest Rutherford conducted an experiment that showed that all the positive charges were concentrated at the center of an atom, and not spread out as Thomson proposed. Rutherford called the concentrated positive charge the nucleus of an atom and compared the electrons that swirled around the nucleus to planets in orbit around the Sun. Both Thompson and Rutherford received Nobel prizes for their work, and Thompson was even knighted – Sir Joseph.
 Thomson and Rutherford c. 1930Electrons were found to have a negative electric charge, and since atoms were neutral, the nucleus must be positive. That was later found to be half right – protons have a positive charge but the nucleus also contains neutrons, which as the name suggests, were neutral, with no charge.
Thomson and Rutherford c. 1930Electrons were found to have a negative electric charge, and since atoms were neutral, the nucleus must be positive. That was later found to be half right – protons have a positive charge but the nucleus also contains neutrons, which as the name suggests, were neutral, with no charge.
Getting back to Rutherford’s model of the atom; the orbiting of the electrons should have produced electromagnetic radiation that would have taken energy from the moving electrons, which then would move closer to the nucleus. Almost immediately the electrons would spiral into the nucleus, destroying the atom. But that obviously doesn’t happen so another model was tossed into the scrap heap of science history. Nobel prizes were not retracted, however.
© 2013 by Wheeling Jesuit University/Center for Educational Technologies®. 316 Washington Ave., Wheeling, WV 26003-6243. All rights reserved. Privacy Policy and Terms of Use.

