-
WHAT IS NASA PHYSICS?
-
MODULES
-
Forces and Motion
-
Conservation of Momentum & Energy
-
Temperature and Heat
-
Fluids
-
Optics
-
Electromagnetic Spectrum
-
Modern Physics
-
Anticipation Guide 7
-
Intro to Modern Physics
-
Blackbody Radiation
-
The Ultraviolet Catastrophe
-
The Photoelectric Effect
-
Bohr's Atom
-
Spectra
-
Radioactive Decay
-
Special Relativity (SR)
-
Simultaneity
-
Distance and Time
-
General Relativity
-
May the Forces be with You
-
Modern Physics Notebook
-
Assessment Problems 7
-
-
Useful Things
-
-
SITE MAP
Temperature and Heat
-
Anticipation Guide
-
Thermal Energy
-
Measuring Temperature
-
Heat Expansion
-
Heat
-
Heat Capacity
-
Change of State
-
Transferring Heat
-
Greenhouse Effect
-
Notebook
-
Assessment Problems
Equation
ΔL = α * L * ΔT
3.7
Ring Thermometer

Do you have your own personal thermometer? A mood ring contains a special fluid made of liquid crystals. The crystals don’t lengthen but change color in response to changing body temperature of your finger (or often of the surrounding air temperature). If your temperature – or the weather’s – changes with your mood the ring would be useful, but it is just a decorative sham.
Image: Wikipedia
The same crystal technology is used in plastic strip thermometers that are calibrated to show a range of about 8° around the standard body temperature of 98.6°F. These are a lot safer than putting a glass tube of poisonous mercury in your mouth as we used to do.
Heat Expansion
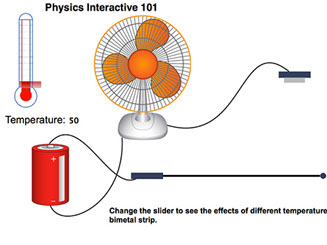
The reason thermometers work is that virtually all materials expand when they are warmed. Liquids expand a lot so it is easy to measure their change, especially in a narrow tube. Metals also expand and they have an important application as a thermal switch. Two strips of metal with different amounts of expansion are bonded together as a bimetallic strip. When heated, one piece of metal expands more than the other, causing the strip to bend. Thermostats in ovens and home furnaces are built around bimetallic strips – when a set temperature is reached the strip bends far enough to make electrical contact, triggering a switch to turn the heat on or off.
Brass and steel are commonly used in bimetallic strips, with brass bending more than steel. The term that quantifies how much a material bends is called the thermal coefficient of linear expansion – whew, a long term – and is represented by the Greek letter α (alpha).
Here are some thermal coefficients of linear expansion, α, for various materials. The units are in millionths per C°.
 Here is how to calculate how much a material changes its length when heated, where Δ means the change of length, L, of temperature, T:
Here is how to calculate how much a material changes its length when heated, where Δ means the change of length, L, of temperature, T:
change in length = alpha x length x change in temperature
ΔL = α * L * ΔT
 Anything constructed of metals – which expand a lot when heated – have to include expansion joints. For example, if during a day a 10 m long iron railway track is warmed 40 C° - as is common in the desert – it will increase its length by:
Anything constructed of metals – which expand a lot when heated – have to include expansion joints. For example, if during a day a 10 m long iron railway track is warmed 40 C° - as is common in the desert – it will increase its length by:
ΔL = α * L * ΔT = 0.000012/C° * 10 m * 40 C° = 0.0048 m = 4.8 mm
If the end of a track touches the next, both will expand, leading to crumpling of the tracks. To minimize this problem tracks have short gaps between them so there is space for expansion. Bridge roadways also expand and so commonly have interlocking vee-shaped metal expansion joints. This is the sort of problem that engineers always have to worry about. Of course, if tracks and bridges were made of diamonds (alpha = 1) their expansion would only be 1/10th as much!
Left: Expansion joint image: CC-BY-SA-3.0/Matt H. Wade at Wikipedia
How Hot are Things?
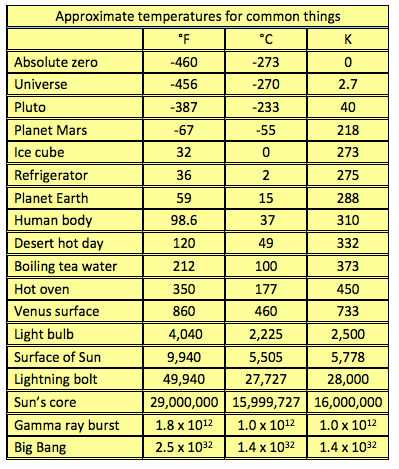 Objects exist with temperatures ranging from nearly absolute zero to billions, even trillions of degrees. Remember that temperature measures the average amount of motion of atoms and molecules but it doesn’t say anything about how many atoms are moving. The temperature may be the same for both a thimble of water and water tower full of it, but there is a huge difference in their internal energy because of the differing sizes of each container.
Objects exist with temperatures ranging from nearly absolute zero to billions, even trillions of degrees. Remember that temperature measures the average amount of motion of atoms and molecules but it doesn’t say anything about how many atoms are moving. The temperature may be the same for both a thimble of water and water tower full of it, but there is a huge difference in their internal energy because of the differing sizes of each container.
© 2013 by Wheeling Jesuit University/Center for Educational Technologies®. 316 Washington Ave., Wheeling, WV 26003-6243. All rights reserved. Privacy Policy and Terms of Use.